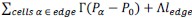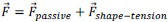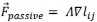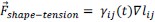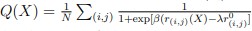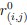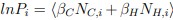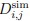Author response:
The following is the authors’ response to the original reviews
eLife Assessment
This important study fills a major geographic and temporal gap in understanding Paleocene mammal evolution in Asia and proposes an intriguing "brawn before bite" hypothesis grounded in diverse analytical approaches. However, the findings are incomplete because limitations in sampling design - such as the use of worn or damaged teeth, the pooling of different tooth positions, and the lack of independence among teeth from the same individuals - introduce uncertainties that weaken support for the reported disparity patterns. The taxonomic focus on predominantly herbivorous clades also narrows the ecological scope of the results. Clarifying methodological choices, expanding the ecological context, and tempering evolutionary interpretations would substantially strengthen the study.
We have now thoroughly revised our manuscript in response to the editor and reviewer’s comments. In particular with regard to:
(1) Sampling design: we clarified our methods section to indicate that we did not use worn or broken teeth in our initial analyses. We added the following sentence around line 690:
“These tooth positions were selected from a broader examination of ~300 individual teeth from 72 specimens. We vetted the specimens and excluded 99 tooth positions (~33% of teeth initially chosen for possible inclusion) from our analyses because they either (1) were partially or completely broken at the crown, (2) were in an advanced stage of attritional wear where no cusps could be identified, or (3) possessed a combination of the two aforementioned conditions.”
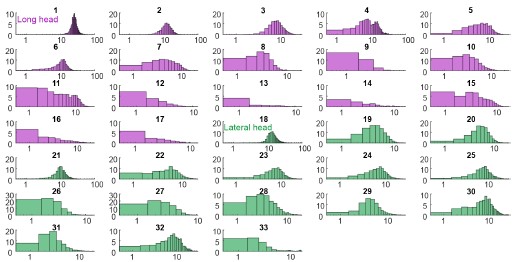
(2) Pooled versus by-tooth position analyses: we repeated the three major analyses (DTA & FEA variability through time, tooth size and variability through time, and DTA-FEA correlation through time) for individual molars (upper M1-3, lower m1-3) and select premolars (upper P3-P4 and lower p4; lower and upper p2 samples contained fewer than 5 specimens across the three time intervals, lower p3 contained only 2 specimens for the middle Paleocene, so they were excluded from the sub-partition analyses).
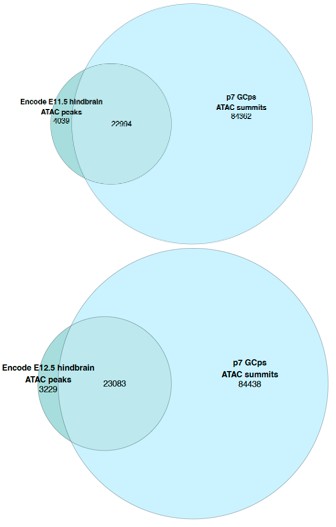
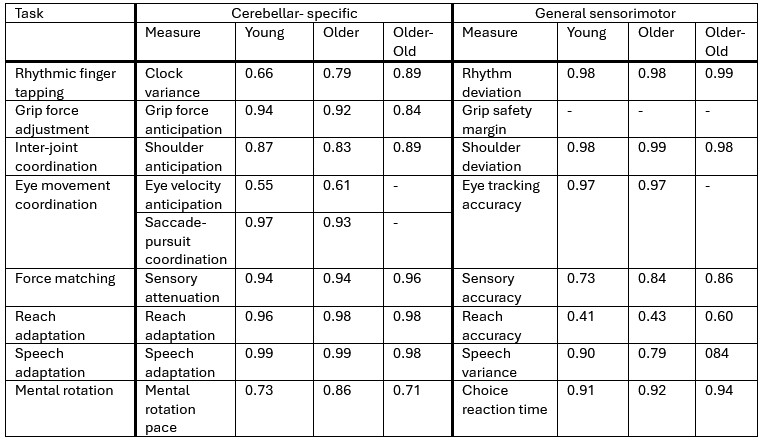
For DTA & FEA variability through time (summarized as a new figure, Fig. S5, also pasted below), OPCR, DNE, and FEA trait data are supported in 78-100% of the per-tooth analyses for both the early-middle Paleocene and middle-late comparisons. By contrast, RFI and Slope data are replicated in only 22-56% of the per-tooth analyses. We qualified the main text reporting and discussion to include these sensitivity analyses so readers can assess nuances in the data when comparing pooled sample versus per-tooth analyses.
For tooth size and variability through time (summarized in a new table, Table S3, also pasted below), we observed broad concordance in the pooled analyses and the per-tooth partitioned analyses. Different tooth positions provide strong support for different aspects of the observed trends, with the lower fourth premolar being the strongest driver of the overall trend. All of the significant trends in per-tooth analyses are in the same direction (i.e., decreasing size disparity and size mean through time) as the pooled sample. We added qualifying clarification in the text to bring attention to these refined results.
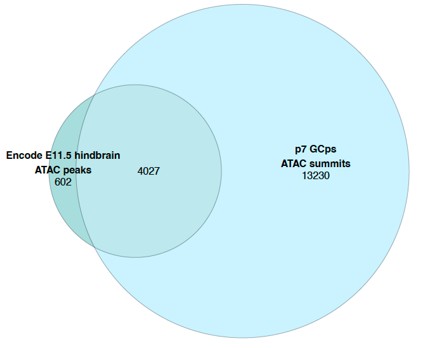
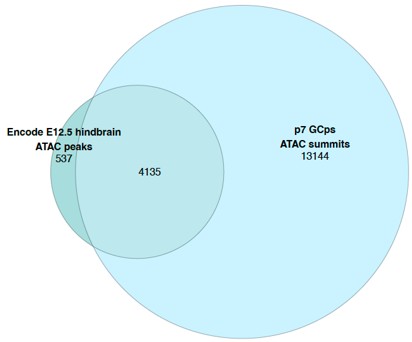
For DTA-FEA correlation through time, we generated per-tooth correlation plots in three new figures (Figs. S9-11, only Fig. S10 shown here as an example). We observed that upper M1 patterns general reflect the trend recovered from analysis of the overall dataset, but M2 and M3 results display inconsistent DTA-FEA correlations, possibly due to small sample sizes. Lower molar patterns generally replicate those recovered in the overall analyses, but lower M1 and M2 signals appear to be stronger than those for lower M3. Finally, low sample sizes make premolar correlations unstable, with general pattern showing EP-MP strengthening then MP-LP stasis or weakening. Given these findings, it appears that the results in the pooled sample correlation plots are mainly driven by lower molar signals. It is not possible to conclude the other tooth position display different patterns because of the limited sample sizes.
(3) Ecological scope of the study: although carnivorans and mesonychids are recorded from some of the time intervals examined in this study, our sampling choice of pantodonts and anagalids reflects the high abundance of available dental specimens in those clades, permitting us to make the strongest statistical inference given the incomplete fossil record. Additionally, all sampled taxa come from archaic clades that have not been determined to be specifically herbivorous; we included an additional paragraph in the introduction to explain this:
“A major challenge with expanding analyses of post K-Pg recovery to Paleocene mammal assemblages elsewhere in the world is the generally stratigraphically limited nature of early Cenozoic sequences. In Asia, Paleocene localities in China represent the best studied to date[11]. From the earliest Paleocene, highly regional and endemic faunas are known from a handful of sedimentary basins (Fig. S1A). Among the faunal elements, only the archaic clades Anagalida and Pantodonta are consistently sampled across the major subdivisions of the Paleocene[11]. An additional complication with ecomorphological analysis of these early mammals is the uncertainty in their dietary ecology, as they are beyond the reach of conventional phylogenetic bracketing approaches to dietary reconstruction. Phenomic analysis of the placental radiation supports insectivory as the ancestral diet of the hypothetical placental ancestor, but uncertainty in the post K-Pg availability of insects and plants in some regions leave some doubt as to the accuracy of this ancestral state reconstruction[1]. Herein we treat the archaic Paleocene taxa in our analyses as having generalized diets rather than categorizing them as insectivores, herbivores, or carnivores.”
Public Reviews:
Reviewer #1 (Public review):
Summary:
This work provides valuable new insights into the Paleocene Asian mammal recovery and diversification dynamics during the first ten million years post-dinosaur extinction. Studies that have examined the mammalian recovery and diversification post-dinosaur extinction have primarily focused on the North American mammal fossil record, and it's unclear if patterns documented in North America are characteristic of global patterns. This study examines dietary metrics of Paleocene Asian mammals and found that there is a body size disparity increase before dietary niche expansion and that dietary metrics track climatic and paleobotanical trends of Asia during the first 10 million years after the dinosaur extinction.
Strengths:
The Asian Paleocene mammal fossil record is greatly understudied, and this work begins to fill important gaps. In particular, the use of interdisciplinary data (i.e., climatic and paleobotanical) is really interesting in conjunction with observed dietary metric trends.
Weaknesses:
While this work has the potential to be exciting and contribute greatly to our understanding of mammalian evolution during the first 10 million years post-dinosaur extinction, the major weakness is in the dental topographic analysis (DTA) dataset.
There are several specimens in Figure 1 that have broken cusps, deep wear facets, and general abrasion. Thus, any values generated from DTA are not accurate and cannot be used to support their claims. Furthermore, the authors analyze all tooth positions at once, which makes this study seem comprehensive (200 individual teeth), but it's unclear what sort of noise this introduces to the study. Typically, DTA studies will analyze a singular tooth position (e.g., Pampush et al. 2018 Biol. J. Linn. Soc.), allowing for more meaningful comparisons and an understanding of what value differences mean. Even so, the dataset consists of only 48 specimens. This means that even if all the specimens were pristinely preserved and generated DTA values could be trusted, it's still only 48 specimens (representing 4 different clades) to capture patterns across 10 million years. For example, the authors note that their results show an increase in OPCR and DNE values from the middle to the late Paleocene in pantodonts. However, if a singular tooth position is analyzed, such as the lower second molar, the middle and late Paleocene partitions are only represented by a singular specimen each. With a sample size this small, it's unlikely that the authors are capturing real trends, which makes the claims of this study highly questionable.
With regard to sampling design: we clarified our methods section to indicate that we did not use worn or broken teeth in our initial analyses. We added the following sentence around line 690:
“These tooth positions were selected from a broader examination of ~300 individual teeth from 72 specimens. We vetted the specimens and excluded 99 tooth positions (~33% of teeth initially chosen for possible inclusion) from our analyses because they either (1) were partially or completely broken at the crown, (2) were in an advanced stage of attritional wear where no cusps could be identified, or (3) possessed a combination of the two aforementioned conditions.”
With regard to pooled versus by-tooth position analyses: we repeated the three major analyses (DTA & FEA variability through time, tooth size and variability through time, and DTA-FEA correlation through time) for individual molars (upper M1-3, lower m1-3) and select premolars (upper P3-P4 and lower p4; lower and upper p2 samples contained fewer than 5 specimens across the three time intervals, lower p3 contained only 2 specimens for the middle Paleocene, so they were excluded from the sub-partition analyses).
For DTA & FEA variability through time (summarized as a new figure, Fig. S5, also pasted below), OPCR, DNE, and FEA trait data are supported in 78-100% of the per-tooth analyses for both the early-middle Paleocene and middle-late comparisons. By contrast, RFI and Slope data are replicated in only 22-56% of the per-tooth analyses. We qualified the main text reporting and discussion to include these sensitivity analyses so readers can assess nuances in the data when comparing pooled sample versus per-tooth analyses.
For the tooth size and variability through time (summarized in a new table, Table S3, also pasted below), we observed broad concordance in the pooled analyses and the per-tooth partitioned analyses. Different tooth positions provide strong support for different aspects of the observed trends, with the lower fourth premolar being the strongest driver of the overall trend. All of the significant trends in per-tooth analyses are in the same direction (i.e., decreasing size disparity and size mean through time) as the pooled sample. We added qualifying clarification in the text to bring attention to these refined results.
For DTA-FEA correlation through time, we generated per-tooth correlation plots in three new figures (Figs. S8-10, only Fig. S9 shown here as an example). We observed that upper M1 patterns general reflect the trend recovered from analysis of the overall dataset, but M2 and M3 results display inconsistent DTA-FEA correlations, possibly due to small sample sizes. Lower molar patterns generally replicate those recovered in the overall analyses, but lower M1 and M2 signals appear to be stronger than those for lower M3. Finally, low sample sizes make premolar correlations unstable, with general pattern showing EP-MP strengthening then MP-LP stasis or weakening. Given these findings, it appears that the results in the pooled sample correlation plots are mainly driven by lower molar signals. It is not possible to conclude the other tooth position display different patterns because of the limited sample sizes.
Reviewer #2 (Public review):
Summary:
This study uses dental traits of a large sample of Chinese mammals to track evolutionary patterns through the Paleocene. It presents and argues for a 'brawn before bite' hypothesis - mammals increased in body size disparity before evolving more specialized or adapted dentitions. The study makes use of an impressive array of analyses, including dental topographic, finite element, and integration analyses, which help to provide a unique insight into mammalian evolutionary patterns.
Strengths:
This paper helps to fill in a major gap in our knowledge of Paleocene mammal patterns in Asia, which is especially important because of the diversification of placentals at that time. The total sample of teeth is impressive and required considerable effort for scanning and analyzing. And there is a wealth of results for DTA, FEA, and integration analyses. Further, some of the results are especially interesting, such as the novel 'brawn before bite' hypothesis and the possible link between shifts in dental traits and arid environments in the Late Paleocene. Overall, I enjoyed reading the paper, and I think the results will be of interest to a broad audience.
Weaknesses:
I have four major concerns with the study, especially related to the sampling of teeth and taxa, that I discuss in more detail below. Due to these issues, I believe that the study is incomplete in its support of the 'brawn before bite' hypothesis. Although my concerns are significant, many of them can be addressed with some simple updates/revisions to analyses or text, and I try to provide constructive advice throughout my review.
(1) If I understand correctly, teeth of different tooth positions (e.g., premolars and molars), and those from the same specimen, are lumped into the same analyses. And unless I missed it, no justification is given for these methodological choices (besides testing for differences in proportions of tooth positions per time bin; L902). I think this creates some major statistical concerns. For example, DTA values for premolars and molars aren't directly comparable (I don't think?) because they have different functions (e.g., greater grinding function for molars). My recommendation is to perform different disparity-through-time analyses for each tooth position, assuming the sample sizes are big enough per time bin. Or, if the authors maintain their current methods/results, they should provide justification in the main text for that choice.
With regard to pooled versus by-tooth position analyses: we repeated the three major analyses (DTA & FEA variability through time, tooth size and variability through time, and DTA-FEA correlation through time) for individual molars (upper M1-3, lower m1-3) and select premolars (upper P3-P4 and lower p4; lower and upper p2 samples contained fewer than 5 specimens across the three time intervals, lower p3 contained only 2 specimens for the middle Paleocene, so they were excluded from the sub-partition analyses).
For DTA & FEA variability through time (summarized as a new figure, Fig. S5, also pasted below), OPCR, DNE, and FEA trait data are supported in 78-100% of the per-tooth analyses for both the early-middle Paleocene and middle-late comparisons. By contrast, RFI and Slope data are replicated in only 22-56% of the per-tooth analyses. We qualified the main text reporting and discussion to include these sensitivity analyses so readers can assess nuances in the data when comparing pooled sample versus per-tooth analyses.
For the tooth size and variability through time (summarized in a new table, Table S3, also pasted below), we observed broad concordance in the pooled analyses and the per-tooth partitioned analyses. Different tooth positions provide strong support for different aspects of the observed trends, with the lower fourth premolar being the strongest driver of the overall trend. All of the significant trends in per-tooth analyses are in the same direction (i.e., decreasing size disparity and size mean through time) as the pooled sample. We added qualifying clarification in the text to bring attention to these refined results.
For DTA-FEA correlation through time, we generated per-tooth correlation plots in three new figures (Figs. S8-10, only Fig. S9 shown here as an example). We observed that upper M1 patterns general reflect the trend recovered from analysis of the overall dataset, but M2 and M3 results display inconsistent DTA-FEA correlations, possibly due to small sample sizes. Lower molar patterns generally replicate those recovered in the overall analyses, but lower M1 and M2 signals appear to be stronger than those for lower M3. Finally, low sample sizes make premolar correlations unstable, with general pattern showing EP-MP strengthening then MP-LP stasis or weakening. Given these findings, it appears that the results in the pooled sample correlation plots are mainly driven by lower molar signals. It is not possible to conclude the other tooth position display different patterns because of the limited sample sizes.
Also, I think lumping teeth from the same specimen into your analyses creates a major statistical concern because the observations aren't independent. In other words, the teeth of the same individual should have relatively similar DTA values, which can greatly bias your results. This is essentially the same issue as phylogenetic non-independence, but taken to a much greater extreme.
It seems like it'd be much more appropriate to perform specimen-level analyses (e.g., Wilson 2013) or species-level analyses (e.g., Grossnickle & Newham 2016) and report those results in the main text. If the authors believe that their methods are justified, then they should explain this in the text.
Based on the per-tooth partition analyses we performed and reported above, the results now show that the overall trends described in the previous draft of the study is a composite of signals from different regions of the dentition. For example, the OPCR, DNE, and FEA trends persist across most tooth positions, whereas the Slope and RFI trends are mainly driven by lower fourth premolar patterns. The tooth size results are also mainly driven by lower fourth premolar patterns, but tooth disparity trends are broadly supported across tooth positions. These observations indicate that the overall trends remain valid, but there are nuances as to which tooth positions are driving which components of the trends. As such, we deem the overall results to be valid, and focused our revision on providing the nuances so readers can assess through-time patterns in more detail than in the previous version of the study.
(2) Maybe I misunderstood, but it sounds like the sampling is almost exclusively clades that are primarily herbivorous/omnivorous (Pantodonta, Arctostylopida, Anagalida, and maybe Tillodonta), which means that the full ecomorphological diversity of the time bins is not being sampled (e.g., insectivores aren't fully sampled). Similarly, the authors say that they "focused sampling" on those major clades and "Additional data were collected on other clades ... opportunistically" (L628). If they favored sampling of specific clades, then doesn't that also bias their results?
If the study is primarily focused on a few herbivorous clades, then the Introduction should be reframed to reflect this. You could explain that you're specifically tracking herbivore patterns after the K-Pg.
We appreciate the reviewer’s suggestion that our sampling may have focused on putative herbivorous clades more than others. However, at the early stage of placental evolution during the Paleocene, and in particular among the endemic forms we studied from south China, it is unclear to us that such clearcut ecomorphological categories were present amongst the fossil mammals. Thus, we take a more agnostic approach and do not define the dietary categories of the sample taxa (and by extension, those of the unsampled taxa). Although we recognize that representatives of certain clades, such as Carnivora, may be more reasonably interpreted as carnivores/insectivores/omnivores and, in the current context, remains unsampled, we point out the fact that including tooth samples from rare taxa such as carnivores likely would have biased the analyses temporally. Chinese Paleocene carnivores are known only from one of the three time intervals analyzed (representing only a handful of specimens), and so would potentially inflate the disparity in that time interval relative to the others (if dentitions specialized for carnivory is assumed to be present in the Paleocene). To clarify this point, we added a paragraph in the introduction:
“A major challenge with expanding analyses of post K-Pg recovery to Paleocene mammal assemblages elsewhere in the world is the generally stratigraphically limited nature of early Cenozoic sequences. In Asia, Paleocene localities in China represent the best studied to date[11]. From the earliest Paleocene, highly regional and endemic faunas are known from a handful of sedimentary basins (Fig. S1A). Among the faunal elements, only the archaic clades Anagalida and Pantodonta are consistently sampled across the major subdivisions of the Paleocene[11]. An additional complication with ecomorphological analysis of these early mammals is the uncertainty in their dietary ecology, as they are beyond the reach of conventional phylogenetic bracketing approaches to dietary reconstruction. Phenomic analysis of the placental radiation supports insectivory as the ancestral diet of the hypothetical placental ancestor, but uncertainty in the post K-Pg availability of insects and plants in some regions leave some doubt as to the accuracy of this ancestral state reconstruction[1]. Herein we treat the archaic Paleocene taxa in our analyses as having generalized diets rather than categorizing them as insectivores, herbivores, or carnivores.”
(3) There are a lot of topics lacking background information, which makes the paper challenging to read for non-experts. Maybe the authors are hindered by a short word limit. But if they can expand their main text, then I strongly recommend the following:
a) The authors should discuss diets. Much of the data are diet correlates (DTA values), but diets are almost never mentioned, except in the Methods. For example, the authors say: "An overall shift towards increased dental topographic trait magnitudes ..." (L137). Does that mean there was a shift toward increased herbivory? If so, why not mention the dietary shift? And if most of the sampled taxa are herbivores (see above comment), then shouldn't herbivory be a focal point of the paper?
We edited the introduction to say that “We used dental topographical traits as indicators of ecomorphological diversity[28] and examined temporal shifts in tooth crown complexity, curvature, and height and their association with tooth performance in terms of deformation resistance using topographic and simulation analyses.” And also added the following to the methods section, in order to clarify that we are using DTA as a general ecomorphological proxy, and not a direct dietary proxy.
“Overall, we use these DTA traits as indicators of ecomorphological capacity, but do not link them explicitly to dietary categories. The craniodental morphology of archaic placental clades in general have not been demonstrated to share the same structure-function linkages as crown mammals, so the aforementioned linkages between DTA and dietary ecology in extant species only serve as evidence that DTA is a potentially useful ecomorphological proxy, without the application of those DTA-diet relationships to the Paleocene fossil mammal dataset.”
b) The authors should expand on "we used dentitions as ecological indicators" (L75). For non-experts, how/why are dentitions linked to ecology? And, again, why not mention diet? A strong link between tooth shape and diet is a critical assumption here (and one I'm sure that all mammalogists agree with), but the authors don't provide justification (at least in the Introduction) for that assumption. Many relevant papers cited later in the Methods could be cited in the Introduction (e.g., Evans et al. 2007).
We added the following sentence to clarify our usage of tooth crowns as ecomorphological proxies: “Teeth are among the most well-preserved parts of fossil mammals, and the fact that they interface directly with the environment through mastication makes them suitable elements for studying potential ecology-morphology linkages.”
c) Include a better introduction of the sample, such as explicitly stating that your sample only includes placentals (assuming that's the case) and is focused on three major clades. Are non-placentals like multituberculates or stem placentals/eutherians found at Chinese Paleocene fossil localities and not sampled in the study, or are they absent in the sampled area?
We modified the following sentence to indicate our sampling focus on placentals: “Our analyses focused on placental mammals from three of the most fossiliferous and biogeographically isolated Paleocene sedimentary sequences in paleotropical Asia: The Nanxiong, Qianshan, and Chijiang Basins in present-day south China 23–27 (Fig. S1)”
d) The way in which "integration" is being used should be defined. That is a loaded term which has been defined in different ways. I also recommend providing more explanation on the integration analyses and what the results mean.
If the authors don't have space to expand the main text, then they should at least expand on the topics in the supplement, with appropriate citations to the supplement in the main text.
We replaced all mentions of “integration” with “covariation” to avoid using the loaded terminology. Covariation more accurately reflects the correlation between two sets of traits (DTA vs FEA) without invoking developmental mechanisms implied by modularity/integration.
(4) Finally, I'm not convinced that the results fully support the 'brawn before bite' hypothesis. I like the hypothesis. However, the 'brawn before ...' part of the hypothesis assumes that body size disparity (L63) increased first, and I don't think that pattern is ever shown. First, body size disparity is never reported or plotted (at least that I could find) - the authors just show the violin plots of the body sizes (Figures 1B, S6A). Second, the authors don't show evidence of an actual increase in body size disparity. Instead, they seem to assume that there was a rapid diversification in the earliest Paleocene, and thus the early Paleocene bin has already "reached maximum saturation" (L148). But what if the body size disparity in the latest Cretaceous was the same as that in the Paleocene? (Although that's unlikely, note that papers like Clauset & Redner 2009 and Grossnickle & Newham 2016 found evidence of greater body size disparity in the latest Cretaceous than is commonly recognized.) Similarly, what if body size disparity increased rapidly in the Eocene? Wouldn't that suggest a 'BITE before brawn' hypothesis? So, without showing when an increase in body size diversity occurred, I don't think that the authors can make a strong argument for 'brawn before [insert any trait]".
Although it's probably well beyond the scope of the study to add Cretaceous or Eocene data, the authors could at least review literature on body size patterns during those times to provide greater evidence for an earliest Paleocene increase in size disparity.
We added a sentence in the discussion of body size during the Paleocene to note that the largest late Cretaceous fossil mammals in China are shrew- to gopher-sized, whereas the largest early Paleocene Chinese Endemic Pantodonts are dog-sized:
“Dog-sized CEPs such as Bemalambda reached sizes not seen in late Cretaceous mammals from China such as Zhangolestes and Kryptobaatar, which are shrew- to gopher-sized [Meng 2014]”
Reference: Meng, J. (2014). Mesozoic mammals of China: implications for phylogeny and early evolution of mammals. Natl. Sci. Rev. 1, 521–542. 10.1093/nsr/nwu070.
Furthermore, we tempered our discussion to restrict the “brawn before bite” hypothesis to post K-Pg recovery in the Paleocene. Body size patterns shifted in the Eocene as crown clades replaced the archaic endemic clades analyzed in our study, and much larger taxa began to appear after the PETM. Such body size shift patterns are based on different clades and likely different dynamics compared to the 10-million year interval examined in our study, so we refrain from commenting on post-Paleocene times.
Recommendations for the authors:
Reviewer #1 (Recommendations for the authors):
(1) In regard to the DTA dataset: Was there a method used to 'fix' these teeth before dental topographic analyses were implemented? If so, this should be explicitly stated. If not, the authors should explain why broken, worn, or abraded teeth were used.
We excluded the incomplete teeth from our analyses. We added the following sentence for clarification: “These tooth positions were selected from a broader examination of ~300 individual teeth from 72 specimens. We vetted the specimens and excluded 99 tooth positions (~33% of teeth initially chosen for possible inclusion) from our analyses because they either (1) were partially or completely broken at the crown, (2) were in an advanced stage of attritional wear where no cusps could be identified, or (3) possessed a combination of the two aforementioned conditions.”
(2) The authors should explicitly explain why all tooth positions were analyzed together. Again, this is not something that is typically done, and some explanation would be helpful for readers.
We added a paragraph in the methods section to explain both our pooled sampling approach, as well as the per-tooth analyses added in this revised manuscript:
“Given the rarity of Paleocene fossil material from China, we combined data from different tooth positions into three pooled samples, one for each of the time intervals examined (early, middle, late Paleocene). We treated the pooled samples as representative of the range of dental topographic features and bite performance traits available to the mammal taxa under study. In this way, the variance estimates are interpreted as measures of the morphological and performance heterogeneity present in each time interval dataset. To further tease out the possibility of specific tooth positions driving the overall trends observed in the pooled samples, we also performed the DTA, FEA, DTA-FEA correlation, and tooth size through-time analyses using per-tooth data partitions.”
(3) I think the authors should hedge their claims a bit more and recognize the limitations of their study (e.g., sample size and tooth preservation).
We thank the reviewer for raising this important point. We carefully read through the main text and further tempered our interpretations based on the limitations of our data. Additionally, we added a paragraph in the supplemental text to summarize the major sources of uncertainty in the sample:
“Sample and methodological limitations
The highly fragmentary nature of early Cenozoic mammal fossils in Asia means that even the best preserved faunas studied herein contain much missing information. First, the absence of a high-resolution chronological framework prevents the fossil data from being analyzed on a continuous time axis; the binning of the samples into three main intervals within a 10-million-year period hinders additional hypotheses about the environmental and climatic correlations of the dental structure-performance results presented. Second, the uneven sampling of the available mammalian assemblage throughout the Paleocene sites in China limits the breadth of ecomorphological categories included in the analyses; rarer taxa representing more specialized carnivore, insectivore, or herbivore forms were not included in our sampling. Third, the spatial discontinuity of stratigraphically younger (Eocene) and older (Cretaceous) mammal assemblages means that body size and ecomorphological shifts bracketing the Paleocene cannot currently be analyzed alongside the dataset presented. These limitations should be taken into account when considering the interpretations made in the main text.”
Reviewer #2 (Recommendations for the authors):
I'm including my Line Comments here as recommendations for the authors. But note that many of my recommendations are also in my Public Review.
L22: "3% of sites"? Do you mean 3% of global sites?
Yes, we revised the sentence to indicate 3% of global sites. Thank you for this suggestion.
L35: This is nitpicky because it's not crucial to your study, but I can't help but point out that the Long Fuse, etc, hypotheses are specifically about the DIVERGENCE TIMES for Placentalia and major subclades, NOT the 'adaptive radiation' of placentals like you imply in your text. Adaptive radiations include ecomorphological diversification and are driven by ecological opportunity (e.g., Schluter 2000). (Emphasis on 'ecological.') The long fuse, short fuse, and explosive models do not include an ecological component - i.e., the diversifications could have occurred without ecological diversification. Instead, for hypotheses that are specifically on the adaptive/ecological radiation of mammals, see the Early Rise, Suppression (or Dinosaur Incumbency; Benevento et al. 2023 Palaeontology), and Late Rise hypotheses (Grossnickle et al. 2019 TREE). These hypotheses apply broadly to all mammals, not just placentals (see Box 1's figure in Grossnickle et al. 2019), but they can still be applied to mammalian subclades like eutherians/placentals (e.g., see Thomas Halliday papers).
Thank you for helping to clarify the adaptive radiation vs. divergence time concepts. We edited this sentence to mention the adaptive radiation hypotheses instead, adding in the references provided by the reviewer.
L39-40: I think your comment is probably accurate. But keep in mind that advocates of the Early Rise and Delayed Rise hypotheses (see citations within Grossnickle et al. 2019) might argue that other time periods, other than the Paleocene, are equally or more important.
We added a reference to Grossnickle et al. 2019 to bring attention to potential arguments otherwise. Thank you for the suggestion.
L48: I think the inclusion of "at higher latitudes" is a little distracting or misleading and should be erased. It implies that the taxonomic diversification was ONLY rapid at higher latitudes. But many of the references that you cite include analyses at the global or continental scale (e.g., Alroy 1999, Grossnickle & Newham 2016) and don't distinguish patterns at different latitudes. If you want to keep the point about latitudes, then I recommend inserting a separate sentence on that point.
We removed “at higher latitudes”.
L50: Isn't "stem lineages and those with no living relatives" somewhat redundant? Or do you mean something like "stem placental/eutherian lineages and extinct placental subgroups"?
Yes, we adopted the suggested phrasing. Thank you.
L53: I recommend starting a new paragraph around here (maybe starting with "Distinct from ...") that focuses specifically on introducing the 'brawn before [ecomorphological trait]' hypothesis.
Done.
L56: "large herbivores and their predators"? Are you just referring to mammals? Wilson (2013), which you cite, and Grossnickle & Newham (2016) argued that dietary specialists were targeted at the K-Pg, but none of the herbivores were "large" (at least relative to Cenozoic herbivores). And most faunivorous mammals at the time were probably insectivorous and not preying on herbivorous mammals, besides maybe a few outlying taxa (e.g., Altacreodus, Nanocuris). I'd revise your sentence for clarity.
We removed “disproportionately impacting large herbivores and their predators” for clarity.
L63: I'd replace "ecometric" with "ecomorphological". Ecometrics commonly refers to using fossil traits to infer paleo environments/climate (e.g., see papers by David Polly, Michelle Lawing, etc), which I don't think is what you're referring to here. (E.g., I don't think that brain size or jaw shape patterns were/are used to infer paleo environments.)
Revised. Thank you.
L85: I strongly advise against making conclusions like this: "Dental height and sharpness variability ... [spiked] in the middle Paleocene corresponding to a short-lived negative excursion in global temperature." That implies that the change in dentitions is linked to global temperature changes, which I don't think your results support. Later in the text you highlight the temporal uncertainty of your time bin ages (L650) and say that the middle Paleocene bin could be as old as ~62 Ma (L646), which is well before the negative excursion (and looks to be more in line with a positive excursion!), at least according to the Figure 1 time scale (see comment below). So, I don't think that your results even support your statement.
We reworded this sentence to say “Dental height and sharpness variability were low in the beginning and end of the time interval, with a peak in the middle Paleocene. This pattern is observed both when dentitions are considered holistically and by tooth position in the lower dentition (Fig. S5; upper teeth display the opposite pattern).”
L144: Using variance for disparity seems fine. But keep in mind that other disparity metrics, such as range (or sum-of-ranges for multivariate data), might produce different results. For instance, variance of RFI and Slope spike in the middle Paleocene, like you point out, but based on the values in Figure 1A, it looks like the ranges stay relatively constant through the Paleocene (although I realize that the ranges might change with bootstrapping). So, your choice of disparity metric might have a big influence on your conclusions. Alternatively, you could calculate disparity using multiple metrics (e.g., Brusatte et al. 2012 Nature Communications; Grossnickle & Newham 2016 supplemental analyses), even if it's just for supplemental analyses.
Thank you for bringing the choice of disparity measures to our attention. We conducted a parallel set of bootstrapped disparity calculation and comparison analyses using range lengths (maximum trait value – minimum trait value for a given trait) and summarized the through-time trends as for variance-based results (Fig. S5). Overall, very similar trends are observed, providing support for the variance-based data interpretation presented in the main text. We added explanation of this additional sensitivity testing both in the main text and in the supplemental text.
L147: "body size disparity ... (Fig. 1B, S6A, Table 1, Data S5)." But I don't see disparity calculated or plotted in any of the figures/tables that you cite. You test for differences in disparity between time bins (Table 1), but that doesn't provide the actual disparity patterns.
We generated a new figure (Fig. S8) to show the tooth size variance and range levels across time and data partitions, and modified this sentence to say that “Over the same time interval examined, body size disparity and mean were higher in the early Paleocene than in subsequent time intervals (Fig. S8, Table S3; also supported by premolar 4 and upper molar partition analyses), indicating that substantial increases in the disparity of dental complexity, curvature, and height lagged behind maximum size disparity tooth size during the Paleocene.”
L151-153: Maybe. But you're basing this on a much narrower temporal range (Paleocene) than the brain and jaw studies, and I think those studies observed big increases in brain/jaw disparity in the Eocene, which you don't sample. And as I explained elsewhere, I'm not convinced that your results strongly support the same pattern. At a minimum, I recommend tempering your conclusions to better reflect the uncertainty of your results.
We tempered our statements here to say that “This suggests a ‘brawn before bite’ pattern in endemic Asian mammals, partially mirroring the endocranial and jaw functional morphology patterns identified in their North American and European counterparts [21,22]. These findings raise the possibility that an initial size-driven post-K-Pg recovery followed by ecomorphological radiation was a global phenomenon, even as regional tectonic events such as the initial collision of the Indian subcontinent with Asia and Deccan Traps volcanism influenced local mammal evolution.”
L170: I'm not well-versed in integration (and modularity) studies, so maybe this reflects my ignorance, but I had trouble understanding sentences like this: "These findings indicate that form-function malleability, the coexistence of distinct topography-performance relationships in each time and taxon partition while overall integration between the two trait groups increases between time bins, was present throughout the Paleocene." If there is space, I recommend revising and/or breaking apart long, jargon-y sentences like that (throughout the paper) so that they're more digestible for readers.
We simplified complex sentences such as the one the reviewer noted, in order to communicate our findings and interpretations more clearly. Thank you for the suggestion.
L183: It's probably fine to assume most placental orders arose in the Paleocene based on fossil evidence. But keep in mind that molecular studies often argue that many orders arose in the Late Cretaceous.
We revised the statement to indicate a “Cretaceous/Paleocene” origin of many modern mammal orders.
L200-207: Again, this might just reflect my ignorance concerning integration analyses, but I recommend expanding on this text to better explain how your integration results support this conclusion. It seems really interesting, and I like the Garden of Eden hypothesis. It's just not immediately clear to me how your results support that hypothesis. A little more background on how to interpret the integration results would be helpful.
We expanded the discussion here to say that “Such flexibility in dental form-function linkage permits ‘mix and match’ trait combinations rather than evolutionary change as a single unit, potentially enhancing the evolvability of feeding ecological traits as new environmental conditions arose [Goswami et al. 2015]”
Reference: Goswami, A., Binder, W.J., Meachen, J., and O’Keefe, F.R. (2015). The fossil record of phenotypic integration and modularity: A deep-time perspective on developmental and evolutionary dynamics. Proc. Natl. Acad. Sci. 112, 4891–4896. 10.1073/pnas.1403667112.
L218: "reached maximum tooth size disparity early". Again, I don't see size disparity plotted or reported. And without baseline comparisons (Late K or Eocene), it's hard to interpret your results and evaluate what 'maximum' means (Figure 1B).
We revised the sentence to now say “In response, Paleocene mammal clades in south China between dental topography and bite performance later, all the while maintaining high levels of variability in dental complexity and convexity (Fig. 1).”
Figure 1A: The time scale in the top left of the figure looks off. Shouldn't the K-Pg be at 66 Ma (not 65 Ma) and the P-E boundary at 56 Ma (not ~54 or 55)?
We revised Fig. 1 to fix the time scale so that K-Pg is at 65.5 Ma and the P-E boundary at 56 Ma. Thank you for catching this.
Figure 1A: Is there a different y-axis scale for the variance (red line) results?
Yes, the y axes for the variance curves were missing. We added them back in. Thank you.
L628-629: As I explained above, it feels like you focused your sampling just on herbivorous/omnivorous groups, and, if true, this is an important point that should be discussed at the forefront of the paper. Does your sample truly represent the total ecological diversity of the mammalian faunas at the time?
We agree with the reviewer about the potential partial sampling of the range of ecomorphological diversity when only the most abundant clades are included in the analyses. However, we refrain from interpreting the dietary groupings represented in the dataset using an assumption of functional morphology from crown/extant clades. We added a paragraph in the introduction to bring attention to the inherent uncertainty in the ecological diversity of the dataset:
“A major challenge with expanding analyses of post K-Pg recovery to Paleocene mammal assemblages elsewhere in the world is the stratigraphically limited nature of early Cenozoic sequences that produce fossil mammals. In Asia, Paleocene localities in China represent the best studied to date 11. From the earliest Paleocene, highly regional and endemic faunas are known from a handful of sedimentary basins (Fig. S1A). Among the faunal elements, only the archaic placental clades Anagalida and Pantodonta are consistently sampled across the major subdivisions of the Paleocene 11. An additional complication with ecomorphological analysis of these early mammals is the uncertainty in their dietary ecology, as they are beyond the reach of conventional phylogenetic bracketing approaches to dietary reconstruction. Phenomic analysis of the placental radiation supports insectivory as the ancestral diet of the hypothetical placental ancestor, but uncertainty in the post K-Pg availability of insects and plants in some regions leave some doubt as to the accuracy of this ancestral state reconstruction 1. Herein we treat the archaic Paleocene taxa in our analyses as having uncharacterized diets rather than categorizing them as insectivores, herbivores, or carnivores. “
L653: Sorry if this is mentioned elsewhere, but did you avoid using teeth with especially worn or broken cusps? You might expand on how you chose teeth for your sample.
We left out this detail in the original submission. Thank you for pointing this out. We had to exclude a third of the teeth because they were too worn or broken. We added the following explanation to the methods section:
“These tooth positions were selected from a broader examination of ~300 individual teeth from 72 specimens. We vetted the specimens and excluded 99 tooth positions (~33% of teeth initially chosen for possible inclusion) from our analyses because they either (1) were partially or completely broken at the crown, (2) were in an advanced stage of attritional wear where no cusps could be identified, or (3) possessed a combination of the two aforementioned conditions.”
L654: "specimens" should be "teeth", correct? In the preceding sentence, you say that there are 200 teeth from only 48 specimens.
Corrected.